 The combined effects of enthalpy and entropy are often combined in what is called "free energy." Free energy is just a way to keep track of the sum of the two effects. 
How much does the enthalpy decrease? How much does the entropy decrease? If the effect of the enthalpy decrease is greater than that of the entropy decrease, the reaction may still go forward. For example, what if enthalpy decreases, but so does entropy? Does the reaction happen, or doesn't it? In that case, we may need quantitation to make a decision. Having two factors may lead to complications. In this case, the red squares will remain just as they are. Both factors tilt the balance of the reaction to the left. If we think of the balance between these two factors, we come to another simple conclusion. In this case, the red squares will be converted into green circles.Īlternatively, maybe entropy decreases when the red squares turn into green circles, and enthalpy increases. Both factors tilt the balance of the reaction to the right. If we think of the balance between these two factors, we come to a simple conclusion. In one case, maybe entropy increases when the red squares turn into green circles, and the enthalpy decreases. Whether or not the reaction proceeds to the right depends on the balance between enthalpy and entropy. Red squares are being converted to green circles, provided the reaction proceeds from left to right as shown. Reactions can happen when entropy increases.Ĭonsider the cartoon reaction below. A reaction is favored if entropy increases: There is also a bias in nature toward increasing entropy in a system.Reactions can happen when enthalpy is transferred to the surroundings. A reaction is favored if enthalpy decreases: There is a bias in nature toward decreasing enthalpy in a system. 
Entropy has something to do with how that energy is stored. Enthalpy has something to do with the energetic content of a system or a molecule. 
Since S = 0 corresponds to perfect order.\)Įntropy and enthalpy are two of the basic factors of thermodynamics. The entropy of a pure crystalline substance at absolute zero (i.e. Nonetheless, the combination of these two ideals constitutes the basis for the third law of thermodynamics: the entropy of any perfectly ordered, crystalline substance at absolute zero is zero. In practice, absolute zero is an ideal temperature that is unobtainable, and a perfect single crystal is also an ideal that cannot be achieved. Such a state of perfect order (or, conversely, zero disorder) corresponds to zero entropy. The only system that meets this criterion is a perfect crystal at a temperature of absolute zero (0 K), in which each component atom, molecule, or ion is fixed in place within a crystal lattice and exhibits no motion (ignoring quantum effects). 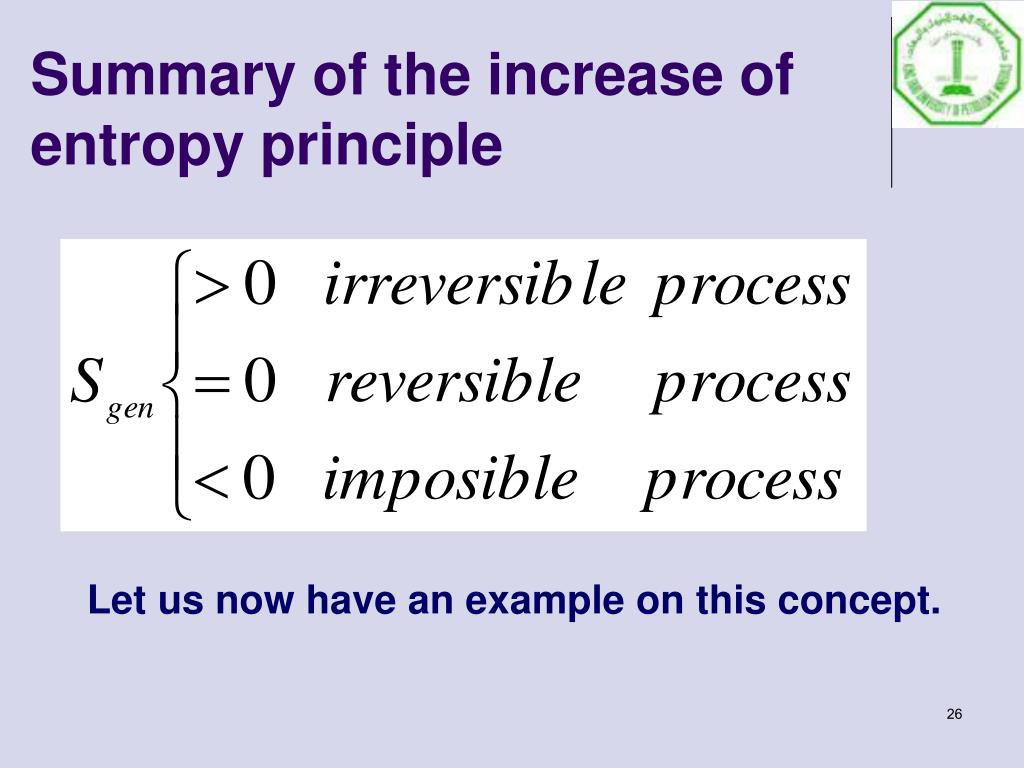
A perfectly ordered system with only a single microstate available to it would have an entropy of zero. The greater the molecular motion of a system, the greater the number of possible microstates and the higher the entropy. These forms of motion are ways in which the molecule can store energy. due to the reduction in the degrees of freedom, the system is more ordered after the reaction).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
